- Polytech
- EMNS
-
Share this page
Transmembrane Transport Activity and Selectivity of Amide-Based Macrocyclic Anion Receptors of Different Sizes
Published on April 8, 2026
– Updated on April 8, 2026
A new article by Hasna, Lau, and Karolis has been published in EurJoc.
by Karolis Norvaisa, Hasna Sherin Ummat, Lau Halgreen, Giulia Piras, Alice Giaprakis, Alessio Cataldo, Stephen J. Butler, Giacomo Picci, Claudia Caltagirone, Hennie Valkenier
First pubished: 7 April. 2026
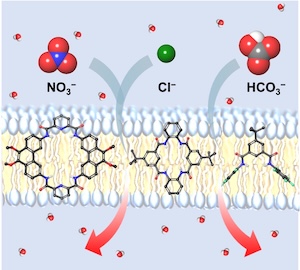 Synthetic anion receptors can transport anions across lipid bilayer membranes. Many anion transporters have been reported, including macrocyclic compounds. However, there are no systematic studies on how the size of macrocycles impacts their anion transport activity and selectivity. Therefore, we prepared eight amide-based macrocyclic compounds with ring sizes ranging from 18 to 26 atoms, as well as four bis-amides. We studied these compounds as anion receptors using chloride titrations and molecular modelling, and as anion transporters using liposomes with various fluorescent probes. Neither the size of the macrocycles nor the affinity for chloride was found to be a determining factor for chloride transport activity; pre-organisation appears to play a more important role. Fluorination was found to have a clear positive effect on anion transport rates, with the bis-amides performing surprisingly well compared to the macrocycles. The smallest non-fluorinated macrocycle exhibited selectivity for chloride over hydroxide and nitrate, whereas the pentafluorophenyl bis-amide demonstrated effective transport of bicarbonate and nitrate, likely due to anion–π interactions in addition to hydrogen bonding. The insights from this study will shape the design of future anionophores.
Synthetic anion receptors can transport anions across lipid bilayer membranes. Many anion transporters have been reported, including macrocyclic compounds. However, there are no systematic studies on how the size of macrocycles impacts their anion transport activity and selectivity. Therefore, we prepared eight amide-based macrocyclic compounds with ring sizes ranging from 18 to 26 atoms, as well as four bis-amides. We studied these compounds as anion receptors using chloride titrations and molecular modelling, and as anion transporters using liposomes with various fluorescent probes. Neither the size of the macrocycles nor the affinity for chloride was found to be a determining factor for chloride transport activity; pre-organisation appears to play a more important role. Fluorination was found to have a clear positive effect on anion transport rates, with the bis-amides performing surprisingly well compared to the macrocycles. The smallest non-fluorinated macrocycle exhibited selectivity for chloride over hydroxide and nitrate, whereas the pentafluorophenyl bis-amide demonstrated effective transport of bicarbonate and nitrate, likely due to anion–π interactions in addition to hydrogen bonding. The insights from this study will shape the design of future anionophores.
First pubished: 7 April. 2026
 Synthetic anion receptors can transport anions across lipid bilayer membranes. Many anion transporters have been reported, including macrocyclic compounds. However, there are no systematic studies on how the size of macrocycles impacts their anion transport activity and selectivity. Therefore, we prepared eight amide-based macrocyclic compounds with ring sizes ranging from 18 to 26 atoms, as well as four bis-amides. We studied these compounds as anion receptors using chloride titrations and molecular modelling, and as anion transporters using liposomes with various fluorescent probes. Neither the size of the macrocycles nor the affinity for chloride was found to be a determining factor for chloride transport activity; pre-organisation appears to play a more important role. Fluorination was found to have a clear positive effect on anion transport rates, with the bis-amides performing surprisingly well compared to the macrocycles. The smallest non-fluorinated macrocycle exhibited selectivity for chloride over hydroxide and nitrate, whereas the pentafluorophenyl bis-amide demonstrated effective transport of bicarbonate and nitrate, likely due to anion–π interactions in addition to hydrogen bonding. The insights from this study will shape the design of future anionophores.
Synthetic anion receptors can transport anions across lipid bilayer membranes. Many anion transporters have been reported, including macrocyclic compounds. However, there are no systematic studies on how the size of macrocycles impacts their anion transport activity and selectivity. Therefore, we prepared eight amide-based macrocyclic compounds with ring sizes ranging from 18 to 26 atoms, as well as four bis-amides. We studied these compounds as anion receptors using chloride titrations and molecular modelling, and as anion transporters using liposomes with various fluorescent probes. Neither the size of the macrocycles nor the affinity for chloride was found to be a determining factor for chloride transport activity; pre-organisation appears to play a more important role. Fluorination was found to have a clear positive effect on anion transport rates, with the bis-amides performing surprisingly well compared to the macrocycles. The smallest non-fluorinated macrocycle exhibited selectivity for chloride over hydroxide and nitrate, whereas the pentafluorophenyl bis-amide demonstrated effective transport of bicarbonate and nitrate, likely due to anion–π interactions in addition to hydrogen bonding. The insights from this study will shape the design of future anionophores.
